Hydraulic-Electric Analogies, Part 1
This file type includes high resolution graphics and schematics when applicable.
Most technical people working in the fluid power industry have roots either in the mechanical realm or the electrical realm. Those who consider themselves “mechanically inclined” often struggle with understanding of electrical concepts. Likewise, those well versed in electrical science are often faced with terminology in fluid power that is inconsistent with their knowledge.
One of the most fundamental examples is a simple on/off switch. An electrical switch blocks flow of electricity when it is open. However, a hydraulic switch (valve) passes flow of a fluid when it is open. In line with this, an electrical switch passes flow when it is closed, whereas a hydraulic or pneumatic valve blocks flow when it is closed. There is a scientifically sound reason why the two terms are used, although there has been some corruption in the electrical case. This topic will be addressed in a future issue.
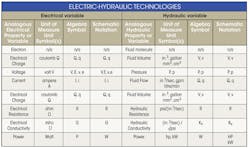
The best place to start a discussion on electric-hydraulic analogies is with the definitions and units of the basic variables, which are given in Table 1. The analogous pairs are voltage and pressure; electrical charge and fluid volume; current and fluid flow; electrical resistance and hydraulic resistance; electrical conductivity and hydraulic conductivity; and power in the two energy transmission media.
Electrons, protons, electrical charge, and fluid molecules
All known material in the universe is comprised of atoms, and each atom has a nucleus with a positive electrical charge. The nucleus is surrounded by orbiting electrons with their negative charge. Under normal circumstances the number of positively charged protons in the nucleus equals the number of orbiting electrons. The number of protons defines the basic material element.
Some elements have electrons that are loosely held by the parent proton, and some have very tightly held electrons. The elements with the most loosely held electrons are metals, and they are the conductors of electricity. The materials with the tightly bound electrons are non-metals. Because they do not conduct electricity, they are insulators.
Even insulators are able to give up their electrons. An example is when you walk across a carpet on a low-humidity day and then touch a metal object — say, a door knob. Just before your finger touches the door knob, you feel a small electrical shock and may see a spark. The reason is that the friction of your insulated shoes gliding across the insulated carpet causes electrons to be forcibly stripped from the carpet and onto your clothing and body. When you reach for the metal door knob, the isolated charges cause the air to ionize, and there is a sudden rush of charges as they attempt to join the metal, and the result is an arc and the shock.
This process is referred to as electrostatics and will not be pursued further, except to state three important observations. First, electrostatic force explains why after combing your hair on a dry day, you can attract small pieces of paper with the comb. Charge separation accounts for the force. Second, this phenomenon is often referred to by lay people as “magnetism.” It is not. It’s an electrostatic phenomenon caused by charge separation. Magnetism is caused by current — that is, electrical charges in motion. Electrostatics is not very important to those practicing electrohydraulics, but magnetism is. Third, to cause the arc between your finger and the door knob and to pick up little pieces of paper with your comb, there must be some force at work in the process. Indeed, there is!
Continuing with the electron-proton model of material, molecules are formed when atoms of different material join together to make entirely new materials that have properties that bear little or no resemblance to the properties of the constituent atoms. There are, literally, unknown numbers of ways in which atoms can combine to form new materials. Of all those ways, only one category of material is important at the moment, namely, hydraulic fluid. Hydraulic fluid is comprised of molecules, and those molecules are to the hydraulic system what electrons are to the electrical system. The fluid molecules do not need an electrical charge to be useful in the hydraulic system, but an electrical charge is vital to all matters electrical.
Voltage, pressure, force, and particle motion
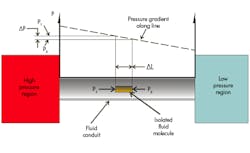
Let’s begin with the hydraulic system by looking at the motivating factor in moving a molecule of fluid around the hydraulic circuit. Figure 1 shows two pressure-containing envelopes, one at a high pressure and the other at a lower pressure. The two envelopes are connected by a fluid conduit, which conducts fluid, but with some resistance. Therefore, a pressure reduction (gradient) exists, as we move from the high-pressure side to the low-pressure side. If a single molecule and the pressures acting on it are isolated, a lower pressure will exist on one side of the molecule than on the other.
Admittedly, molecules are tiny, but no matter how tiny, there will be a difference in pressure from one end to the other. The resulting force acting on the correspondingly tiny area of the molecule will motivate the molecule to move to the right, toward the low-pressure region. This is a complicated way of explaining that fluid tends to move from a region of higher pressure to a region of lower pressure.
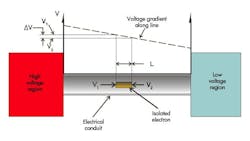
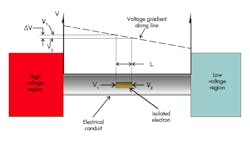
A perfectly analogous situation exists in the electrical circuit. Consider the situation depicted in simplified fashion in Figure 2. But first, consider the unit of voltage.
Voltage is a measure of the potential energy (newton-meter, also called the joule) stored per coulomb of electrical charge. A coulomb of electrical charge is merely 6.241 × 1018 electrons. The voltage gradient that exists along the conductor has a slope of volts per meter. That gradient is called an electric field, usually symbolized as E. When the voltage is multiplied by the value of E, the result has units of newton per coulomb. The isolated electron, with its miniscule electrical charge of 1.602 × 10-19 coulomb, undergoes an equally miniscule, non-zero force. This force motivates the electron to head for the lower voltage region.
When you walk across the carpet on a dry day and reach for the door knob, your body is the high-voltage region, often thousands of volts. However, the door knob is low voltage, and the electric field is so strong that it ionizes the air, turning it into the conductor, and the result is a small bolt of lighting. On a more humid day, two conditions prevent this from happening. First, there is simply less charge transferred to your body, and second, the humidity allows any charges on your body to “leak back” to the carpet through your skin and clothing.
This discussion will continue next month, beginning with how voltage and pressure are measured using voltmeters and pressure gauges.
Jack Johnson is an electrohydraulic specialist, fluid power engineering consultant, and president of IDAS Engineering Inc., Milwaukee. Contact him at [email protected], phone (414) 236-5350, or visit www.idaseng.com.
This file type includes high resolution graphics and schematics when applicable.